Introduction to Systems Biology (Coursera)
Categories
Effort
Languages
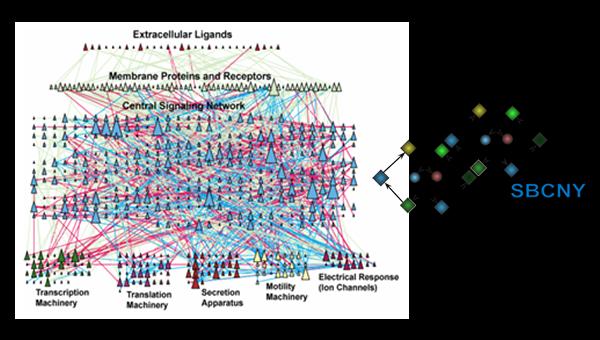
This course will introduce the student to contemporary Systems Biology focused on mammalian cells, their constituents and their functions. Biology is moving from molecular to modular. As our knowledge of our genome and gene expression deepens and we develop lists of molecules (proteins, lipids, ions) involved in cellular processes, [...]
Apr 22nd 2024